Calculate the increase in energy (in joule) per atom of a piece of aluminium when its temperature is raised by 1^(@)C. Given 27 g of aluminium contains 6xx10^(23) atoms, and specific heat

Estimate the average thermal energy of a helium atom at(i) room temperature (27 ^∘C ),(ii) the temperature on the surface of the Sun (6000 K),(iii) the temperature of 10 million kelvin (the
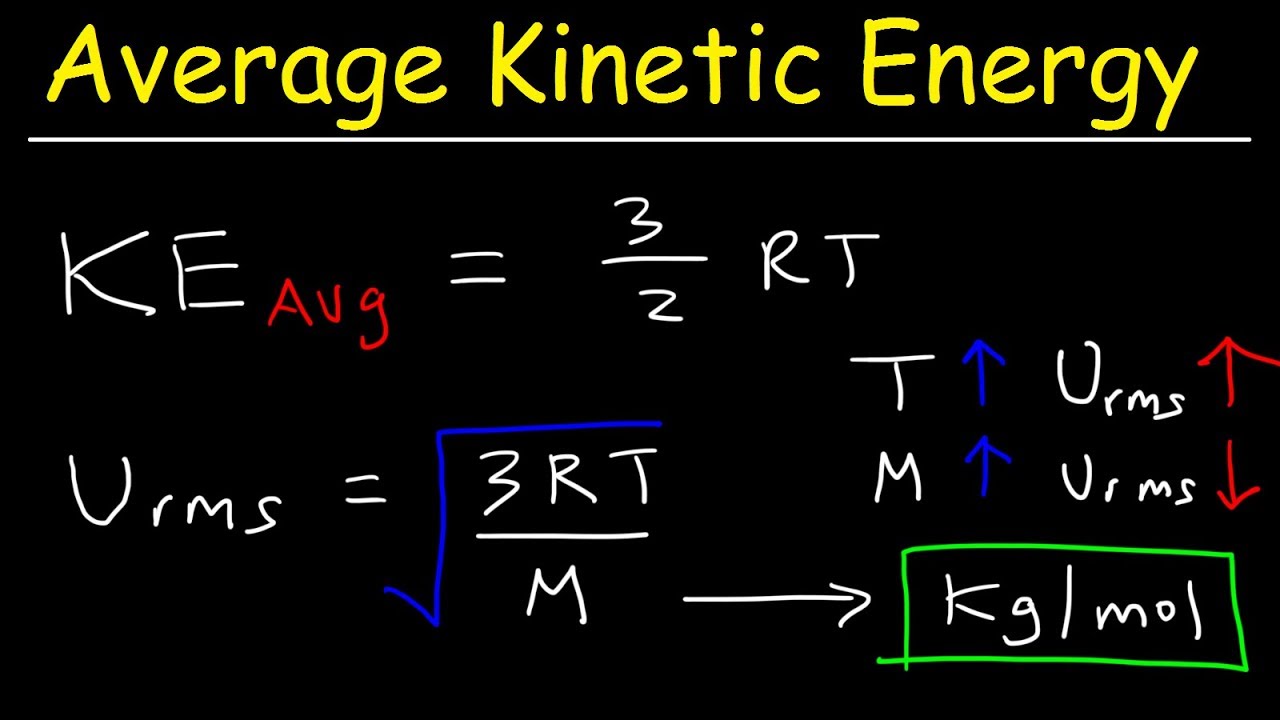
Average Kinetic Energy of a Gas and Root Mean Square Velocity Practice Problems - Chemistry Gas Laws - YouTube

SOLVED:In Part IV you'll learn to calculate that 1 mole (6.02 ×10^23 atoms) of helium atoms in the gas phase has 3700 J of microscopic kinetic energy at room temperature. If we

SOLVED: Calculate the fraction of atom sites that are vacant for lead at its melting temperature of 327*C. Assume an energy for vacancy formation of 0.52 eVlatom: When, Boltznann constant K =
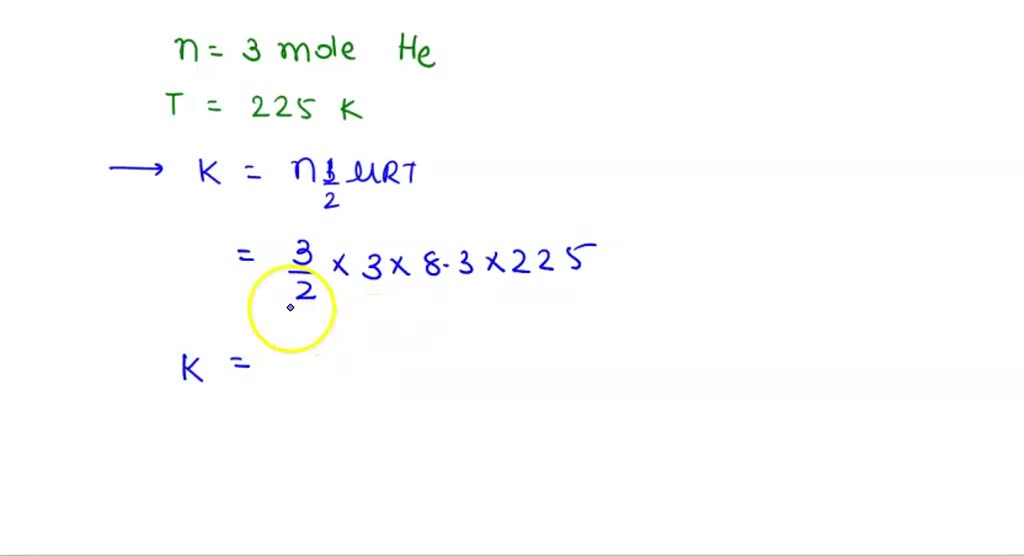
SOLVED: Five moles of a helium gas are at a temperature of 245 K. Calculate the average kinetic energy per atom, the root-mean-square (rms) speed of atoms in the gas, and the

Thermal Energy Equation & Examples | How to Calculate Thermal Energy - Video & Lesson Transcript | Study.com