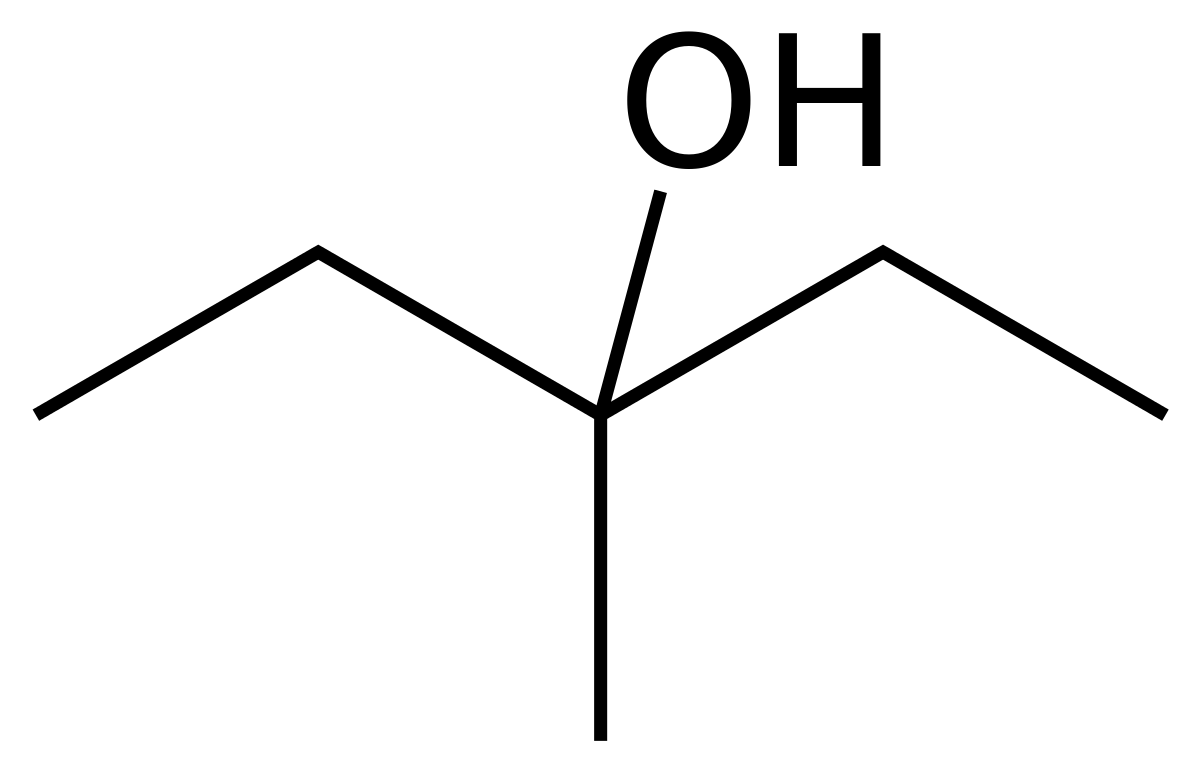
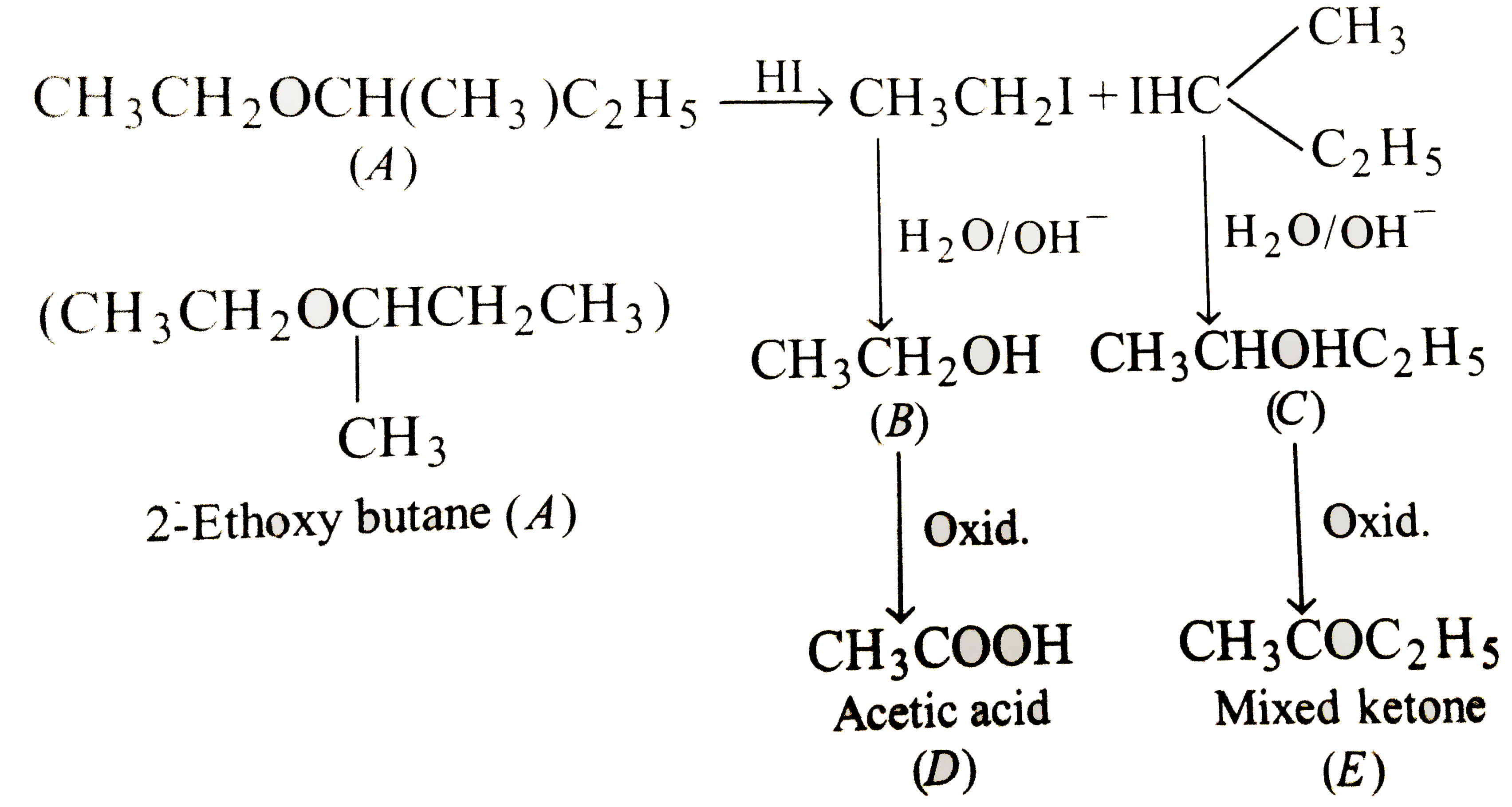Compound X is 2-propanol. When compound X is heated with a strong acid, it dehydrates to compound Y (C_3H_6). When compound X is oxidized, compound Z is forms which cannot be oxidized

PDF) Efficient Catalytic Dehydration of High-Concentration 1-Butanol with Zn-Mn-Co Modified γ-Al2O3 in Jet Fuel Production

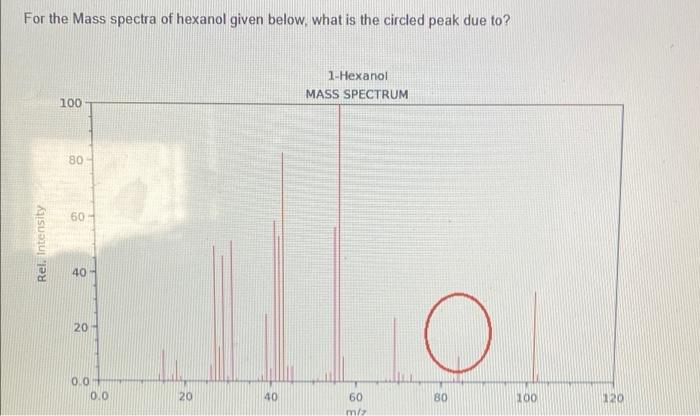
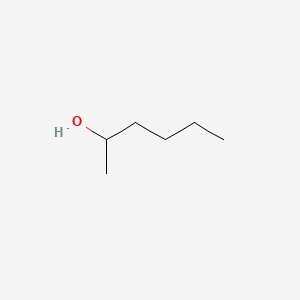
SOLVED: Draw the reactants and products for each of the chemical rcactions below: Dehydration of pure hexanol at 180*€ in the presence of H,SOs Dehydration of a mixture ethanol and [-propanol at

SOLVED: Spccify both the alcohol starting ' matcrial and the reagents you would use in cach stcp in a synthesis of the compound shown: If the synthesis requires only two steps enter

Dehydration of Pinacolyl Alcohol - Dehydration of Pinacolyl Alcohol Introduction H2SO4 H2SO4 C6H14O C6H12 C6H12 Figure 1. Chemical Equation for the | Course Hero
S 1 Reaction Remember that a 3˚ alkyl halide will not undergo a S 2 reaction The steric hindrance in the transition state

Saline-Sodium Citrate (SSC), 20X Solution (Molecular Biology), Fisher BioReagents Nalgene Poly Bottle; 1L Saline-Sodium Citrate (SSC), 20X Solution (Molecular Biology), Fisher BioReagents | Fisher Scientific
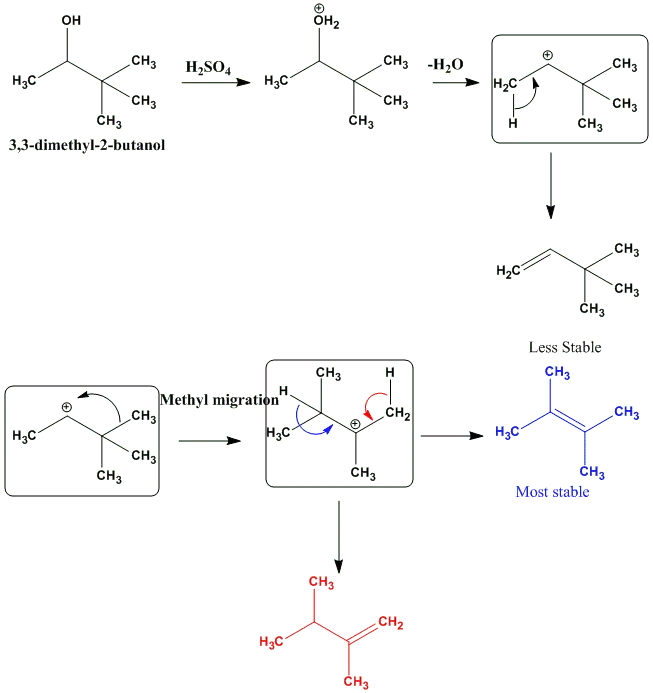
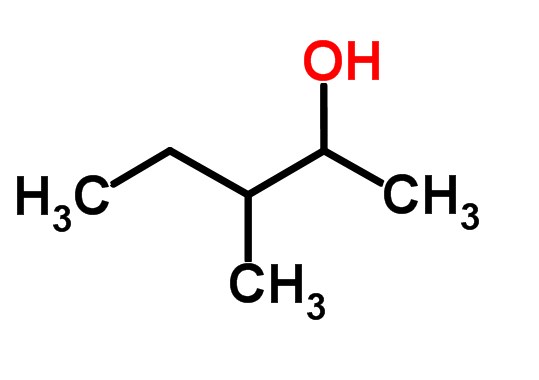
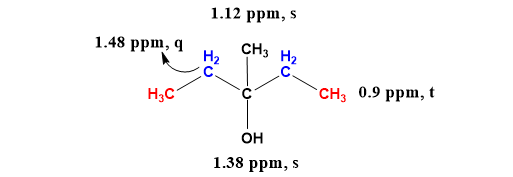
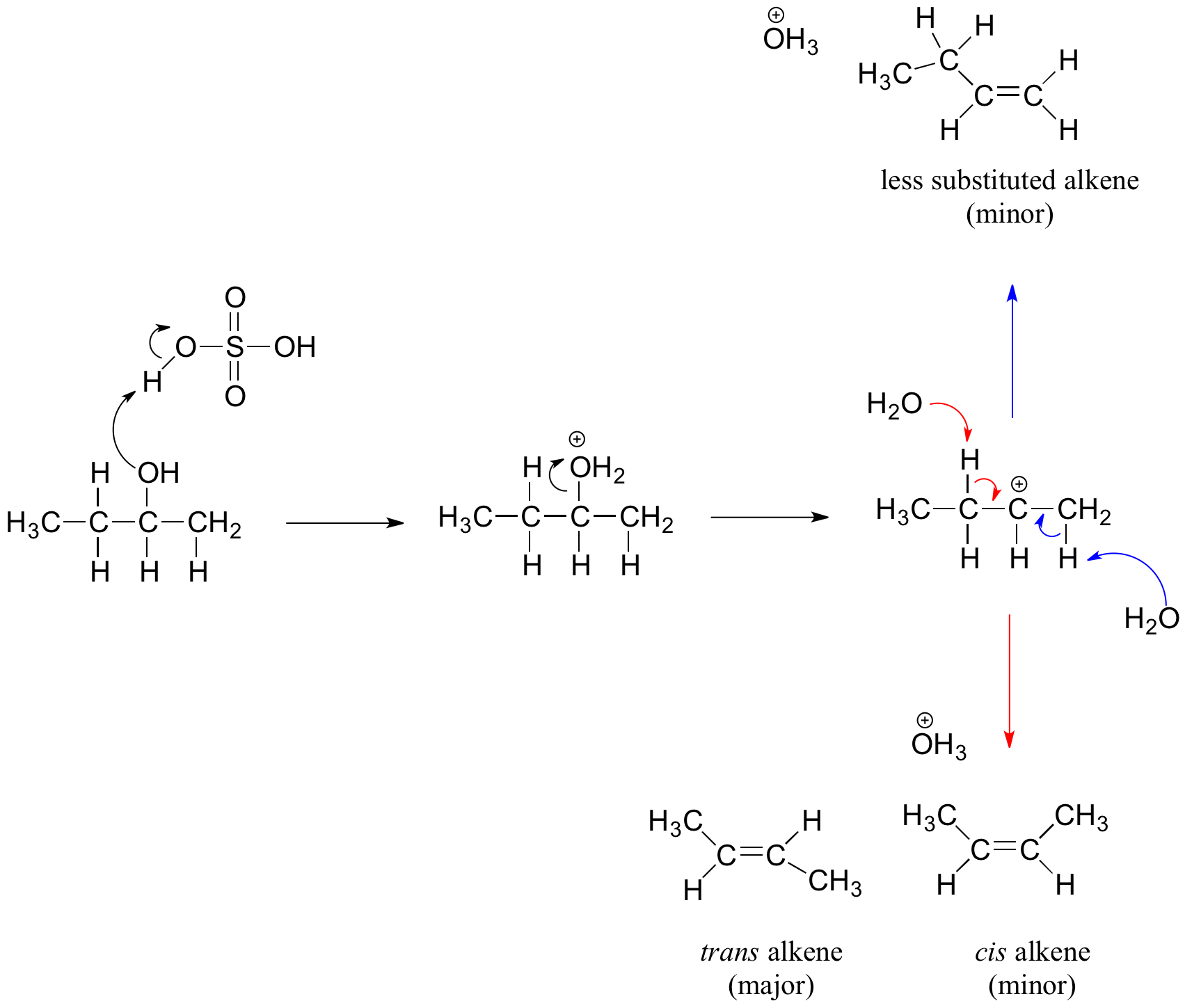
OneClass: Acid-catalyzed dehydration of alcohol 3,3-demethyl-2-butanol, togive three isomeric alkenes...